You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
COVID-19 Coronavirus Thread
- Thread starter country_hick
- Start date
-
- Tags
- chinese virus outbreak
- Status
- Not open for further replies.
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
I can't imagine that many people will want to watch that after seeing the trailer!Lets just hope we dont get to covid - 23 levels.
The Oxford vaccine seems to have missed out Antarctica, but they have completed their trial (not just preliminary results), effective, can be stored in a normal fridge for 6 months, and 3 billion doses on the way!
AZD1222 vaccine met primary efficacy endpoint in preventing COVID-19
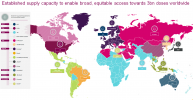
USA not shaded in, along with Greenland.
Last edited:
joe384
Well-Known Member
- Joined
- Aug 29, 2019
- Messages
- 639
- Reaction score
- 301
- Location
- Essex
- Country
- United Kingdom
- Dash Cam
- Viofo A139 3CH, A139 2CH, Mobius w/ Varifocal Zoom
1918 pandemic newspaper headlines. Open-Face Sneezers!!!
Lol I encountered one while paying for my petrol on Saturday, stood right near me as well! Even though we are technically in lockdown in the UK it feels like people aren't really being as careful as back in during our first national lockdown
SawMaster
Well-Known Member
- Joined
- Apr 10, 2015
- Messages
- 9,450
- Reaction score
- 8,318
- Location
- SC
- Country
- United States
- Dash Cam
- Numerous and ever-changing
Oxford tweeted that their vaccine was 70.4% effective which is quite lower than the Pfizer and Moderna versions
https://www.ox.ac.uk/news/2020-11-23-oxford-university-breakthrough-global-covid-19-vaccine
Then they went on to say one dosage method was 90% effective while the other was 63%. Statements like this are confusing and they should know how to word things better than this
Still glad to see any progress toward bringing the pandemic to an end
Phil
https://www.ox.ac.uk/news/2020-11-23-oxford-university-breakthrough-global-covid-19-vaccine
Then they went on to say one dosage method was 90% effective while the other was 63%. Statements like this are confusing and they should know how to word things better than this
Still glad to see any progress toward bringing the pandemic to an end
Phil
kamkar
Well-Known Member
- Joined
- Oct 11, 2013
- Messages
- 34,636
- Reaction score
- 19,060
- Country
- Denmark
- Dash Cam
- 10 years, many dashcams
70% i think are in line with the regular flu vaccines we have been using for a long time, i would also take 70% if this was a flesh eating flu.
Price should also be along the lines of regular vaccines, so 10 to 50 USD i think is the norm.
But of course if you need 2 shots, then 100 USD thats pretty expensive for many people.
Also pretty sure someone will be capitalizing on this in a illegal way, but unsure if the people in charge will be prepared for that,,,,,, i somehow doubt that as pro-activeness and prepping are not really the hallmarks or any government i know of.
And i am sure when someone can exploit messed up tax systems in some countries and steal billions from those countries, i am sure filthy animals like that will also have a plan to capitalize on this corona mess.
And these parasites range from individual persons to big banks.
Price should also be along the lines of regular vaccines, so 10 to 50 USD i think is the norm.
But of course if you need 2 shots, then 100 USD thats pretty expensive for many people.
Also pretty sure someone will be capitalizing on this in a illegal way, but unsure if the people in charge will be prepared for that,,,,,, i somehow doubt that as pro-activeness and prepping are not really the hallmarks or any government i know of.
And i am sure when someone can exploit messed up tax systems in some countries and steal billions from those countries, i am sure filthy animals like that will also have a plan to capitalize on this corona mess.
And these parasites range from individual persons to big banks.
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
I think they did it deliberately to avoid doing what Moderna did. There was not enough data in the Moderna study to say if it was 94 or 96 or 89, and although Oxford has more data, having tried multiple dosing regimes they have even less data for the 90% claim, so they have rounded down to the nearest ten for an estimate for each regime, I think excluded regimes with less data, and used the average of 70 for the entire study since that is the only number with enough data to be statistically proven at the current time. It is probably even worse if you look at the detail, and the same is true of Moderna which only had 11 people get ill during the study, which doesn't make for accurate predictions to fractions of a %!Statements like this are confusing and they should know how to word things better than this
The data will improve over time, and it is good to see that they experimented to find the best dosing strategy even though that made their overall result worse.
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
This is for protection against infection, protection against getting ill was 100% - much better than flu!70% i think are in line with the regular flu vaccines
no hospitalisations or severe cases of the disease were reported in participants receiving the vaccine.
SawMaster
Well-Known Member
- Joined
- Apr 10, 2015
- Messages
- 9,450
- Reaction score
- 8,318
- Location
- SC
- Country
- United States
- Dash Cam
- Numerous and ever-changing
I don't keep up with such things, but you can bet your backside that investors were buying up the shares for the vaccine makers many many months ago, so they will be reaping some profits. But some of these companies have already pledged to get the vaccine out at their cost or close to that so there won't be any windfalls made on it directly. Given the need for special handling (shipment cooling and limited shelf-life) it won't be very useful if a shipment is stolen, even moreso when you need two doses over time which will probably be coming from two separate shipments. And the shipments and distribution will be very closely monitored.
But as always there will be "self-important" people getting some of the first doses like politicians and hospital administrators simply because they are the ones making the distribution arrangements. And same as always those who cannot afford to pay for it will be the last to get it. Some things never change although they should
Moderna's initial statement was clearer than this. They claimed efficacy at the dosage regimen which they planned to be distributing and using, noting immediately that it could be a few percentage points off as could be anyone;'s claims. They did explain the two different dosage regimens tested and the results of each but didn't include the lesser-effective rates because that would not be applicable since it wasn't going to be used. Some nutter is going to come along and claim that Oxford/Astrazeneca is not as effective and that there's some great conspiracy involved which is designed to enslave and zombify the world using just the first part of this 'tweet', ignoring the rest
Again some things never change, but at least things like this can be better dealt with by more careful choosing of the words you use and how you use them.
Phil
But as always there will be "self-important" people getting some of the first doses like politicians and hospital administrators simply because they are the ones making the distribution arrangements. And same as always those who cannot afford to pay for it will be the last to get it. Some things never change although they should
Moderna's initial statement was clearer than this. They claimed efficacy at the dosage regimen which they planned to be distributing and using, noting immediately that it could be a few percentage points off as could be anyone;'s claims. They did explain the two different dosage regimens tested and the results of each but didn't include the lesser-effective rates because that would not be applicable since it wasn't going to be used. Some nutter is going to come along and claim that Oxford/Astrazeneca is not as effective and that there's some great conspiracy involved which is designed to enslave and zombify the world using just the first part of this 'tweet', ignoring the rest
Again some things never change, but at least things like this can be better dealt with by more careful choosing of the words you use and how you use them.
Phil
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
Yes, and that allows them to reply that 3 billion doses that can be shipped anywhere in the world that has a normal refrigerator is far more effective than Moderna's few million that need special storage and transport, even if Moderna's turns out to be a few % more effective, and there is currently not enough data to say that Moderna's is statistically more effective. It maybe that Oxford has an even better dosing regime under test but doesn't currently have enough data to prove the 97% effectiveness, and overtaking Moderna in a few months time will create great headlines!Some nutter is going to come along and claim that Oxford/Astrazeneca is not as effective
We will find out over time, but currently there is no real difference between 90 and 94 given the amount of data collected, it could go either way, or more likely they are both doing the same thing with the exact same result!
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
Astra Covid-Shot Data Leaves Some Analysts Questioning It
Results from a crucial study of AstraZeneca Plc and the University of Oxford’s Covid-19 vaccine drew a harsh review from at least one sell-side analyst as Wall Street grappled with the future of a potentially less effective shot.
The results showed the vaccine stopped an average of 70% of patients from falling sick. However, the company’s formatting of the data that highlighted a 90% effectiveness drew skepticism from SVB Leerink analyst Geoffrey Porges. The analyst said the company highlighted results from a “relatively small” group of volunteers in the trial and wouldn’t get U.S. approval based on a lack of diversity among participants, he wrote in a note.
Porges wasn’t alone on Wall Street in questioning the results from Astra and Oxford. Jefferies analyst Michael Yee called the data “mixed” and highlighted that results from competitors including Pfizer Inc. and partner BioNTech SE as well as Moderna Inc. looked more robust.
While the Astra and Oxford program had positive effectiveness, Yee questioned which countries would prefer to use the vaccine that has notably lower efficacy. “Having any cases of Covid implies big risk, so why not use the best (vaccines), and which populations of citizens would be OK knowing they are getting one that has notably lower efficacy?” Yee asked.
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
Of course Wall street people don't want anybody buying this one, it is being sold at cost, so there are no profits to be made, whereas Moderna is selling at a huge profit!
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
Of course Wall street people don't want anybody buying this one, it is being sold at cost, so there are no profits to be made, whereas Moderna is selling at a huge profit!
They are questioning the way the data was presented regarding the 90% claim as well as the lower efficacy overall. As the analyst points out the small sample data presented so far for the 90% efficacy claim will not pass muster with US regulators.
Of course, we can surely expect plenty of Nigelsplaining over this one after months of empty boasting about the (British) Oxford vaccine along with constant unfounded criticism of Moderna and Pzifer's approach.
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
The 70% is enough to get USA approval, except that they needed 30,000 participants and an extended timescale for the USA regulator, which they don't have yet for any of the figures. Meanwhile in the rest of the world, there are sufficient numbers for the regulators and the stock price isn't the most important figure of all - it is not being sold for profit, but that is a worry for Wall Street and Fauci who have been seeing their Moderna stocks skyrocket, but which now may tumble if the rest of the world turns out to be uninterested in purchasing the vastly more expensive and difficult to transport Moderna vaccine.As the analyst points out the small sample data presented so far for the 90% efficacy claim will not pass muster with US regulators.
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
The 70% is enough to get USA approval, except that they needed 30,000 participants and an extended timescale for the USA regulator, which they don't have yet for any of the figures. Meanwhile in the rest of the world, there are sufficient numbers for the regulators and the stock price isn't the most important figure of all - it is not being sold for profit, but that is a worry for Wall Street and Fauci who have been seeing their Moderna stocks skyrocket, but which now may tumble if the rest of the world turns out to be uninterested in purchasing the vastly more expensive and difficult to transport Moderna vaccine.
Here comes the predicted NIGELSPLAINING!
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
Porges also said Astra and Oxford officials would be “roundly criticized” for a safety disclosure that was “hardly reassuring.” The analyst said the company highlighted results from a “relatively small” group of volunteers in the trial and wouldn’t get U.S. approval based on a lack of diversity among participants, he wrote in a note.
Porges wasn’t alone on Wall Street in questioning the results from Astra and Oxford. Jefferies analyst Michael Yee called the data “mixed”.
"While the Astra and Oxford program had positive effectiveness, Yee questioned which countries would prefer to use the vaccine that has notably lower efficacy. “Having any cases of Covid implies big risk, so why not use the best (vaccines), and which populations of citizens would be OK knowing they are getting one that has notably lower efficacy?” Yee asked.
Despite the tumble in AstraZeneca's stock price in London and NY, Goeffrey Porge's company SVB Leerink holds an outperform rating on Astra with a $65 price target.
Porges wasn’t alone on Wall Street in questioning the results from Astra and Oxford. Jefferies analyst Michael Yee called the data “mixed”.
"While the Astra and Oxford program had positive effectiveness, Yee questioned which countries would prefer to use the vaccine that has notably lower efficacy. “Having any cases of Covid implies big risk, so why not use the best (vaccines), and which populations of citizens would be OK knowing they are getting one that has notably lower efficacy?” Yee asked.
Despite the tumble in AstraZeneca's stock price in London and NY, Goeffrey Porge's company SVB Leerink holds an outperform rating on Astra with a $65 price target.
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
You must have bought some Moderna shares, since that was the first thing you checked as soon as you heard the newsHere comes the predicted NIGELSPLAINING!
Dashmellow
Well-Known Member
- Joined
- Sep 22, 2013
- Messages
- 18,813
- Reaction score
- 19,427
- Location
- Uncanny Valley (●_●)
- Country
- United States
- Dash Cam
- Umpteen
You must have bought some Moderna shares, since that was the first thing you checked as soon as you heard the news
No, the stock quotes are from the Bloomberg article I posted.
The way you've been touting Oxford/AstraZeneca since the get-go while criticizing Moderna and other companies, one has to suspect that you hold shares in AstraZeneca. Perhaps there is more to your British ultra-nationalism than appears on the surface?
Nigel
Well-Known Member
- Joined
- Jul 7, 2014
- Messages
- 17,558
- Reaction score
- 9,194
- Location
- Wales
- Country
- United Kingdom
- Dash Cam
- Gitup F1+G3ꞈꞈꞈꞈꞈ Viofo A229ꞈꞈꞈꞈꞈ Blueskysea B4K
"cloth masks were associated with a significantly higher risk", even N95 masks don't work for viruses, just wash your hands!
The "Plain language summary" is the easiest bit to read.
The "Plain language summary" is the easiest bit to read.
- Status
- Not open for further replies.


