Does "thermal runaway (chemical combustion)" mean explosion?
If I do not mind if itshorten the lifespan of the bank's cells and prematurely reduce its charging capacity, can I use a normal powerbank for my purpose?
Since you've asked the exact same question for a second time I will give you the exact same answer for the second time. No.
Charging a lithium-ion battery bank at the same time it is discharging is an unwise idea, period!
The same factors are at work that will shorten the lifespan of the battery bank if you do this are the SAME ones that can create the conditions for it to catch fire or even explode. You cannot simply rationalize that you can tolerate a shorter lifespan for the device without accepting the fact that it also might eventually catch fire in the process. If you abuse lithium-ion batteries you don't get to pick and choose which problem you might encounter.
Despite all the evidence here on the pages of DCT, including in this very thread going back two years now people keep asking if pass through charging is viable and then when they hear an answer they don't like they insist that somehow they can do it anyway as if the laws of physics or electro-chemistry do not apply to them.
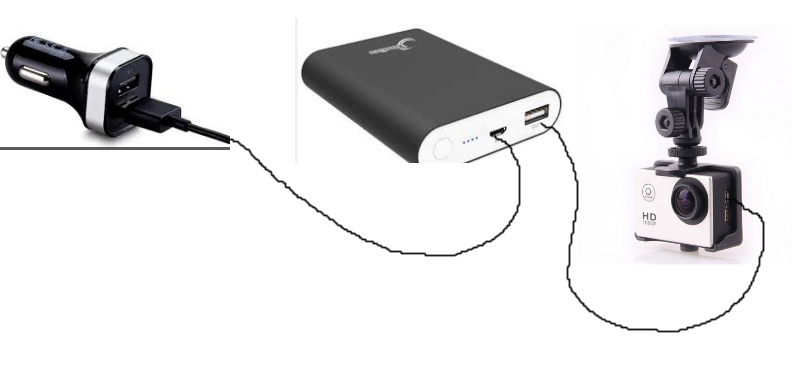
Powerbanks were never designed to be permanent uninterruptible power supplies for dash cams. They are designed for occasional back-up charging of smartphones and tablets or to
temporarily power certain other devices.
While I provided a link to a previous detailed explanation of why lithium-ion batteries are placed under severe stress if they are charged and discharged at the same time I will repeat the explanation here in brief:
When the batteries are charged, electricity drives lithium ions from the cathode, across an ion-filled electrolyte fluid, and into the anode, which is made of stacks of graphite where it stores this energy until it is later released as electrons to power your device. As the battery drains, the lithium ions migrate from the anode back towards the cathode. So, if you try to charge the battery at the same time you are discharging it you are forcing ions to travel in
opposite directions through the electrolyte at the
SAME time and this causes much stress (and heat) within the cells. This stress eventually causes some of the lithium ions in the electrolyte to form metallic lithium (called plating) which is flammable and burns at a very high temperature. While some lithium plating is normal and is part of the aging process of a lithium cell, accelerating this process by placing the cell under undue stress causes problems and compromises safety. This rapid build-up of metallic lithium can under certain circumstances eventually cause a direct short circuit within the battery or it can merely become a source of fuel for a fire caused when the electrolyte chemistry overheats. Once this stress caused overheating process starts it can create a situation where the increase in temperature can change the conditions in such a way that causes yet a further increase in temperature in a chain reaction often leading to a full fledged fire that ignites the metallic lithium that has been forming. What can happen then is that hot volatile gases are released when the battery swells and finally bursts. Once the battery cell bursts these hot gases and burning lithium metal very suddenly come into contact with oxygen and this leads to a catastrophic result. That is what is meant by the term "thermal runaway" in regard to lithium-ion batteries.
Does "thermal runaway (chemical combustion)" mean explosion?
A "thermal runaway" as described above causes a fire that can't easily be stopped until all the fuel is used up. Usually, that will mean just a fire like in all those Samsung Galaxy Note 7 that were in the news recently after catching fire when their batteries failed. However, smartphones and dash cameras use a form of lithium-ion battery called a lithium-polymer which is basically a lithium battery in a foil wrapper. If these foil wrapped batteries have a problem most of the time they will simply swell up. If they burst and catch fire the expanding gases can easily escape.
Powerbanks are not the same. They are usually made with individual 18650 lithium-ion battery cells. A typical 20,000 mAh powerbank uses eight 25o0 mAh 18650 cells. These are sealed metal cylinders in the traditional shape of a typical alkaline cell. Unlike the foil pouched lithium-polymer battery you will find in a typical dash cam or smart phone these 18650 cells are not able to expand and release hot gases during a thermal runway and when enough pressure builds up inside them they can indeed explode. Of course, this will all be happening to the battery cells inside the sealed housing of the power bank thus compounding matters. Think hand grenade.
There are still
other reasons not to charge while discharging. Isidor Buchmann, the battery scientist behind
Battery University explains:
"A portable device must be turned off during charge. This allows the battery to reach the set threshold voltage unhindered, and enables terminating charge on low current. A parasitic load confuses the charger by depressing the battery voltage and preventing the current in the saturation stage to drop low. A battery may be fully charged, but the prevailing conditions prompt a continued charge. This causes undue battery stress and compromises safety."
Aside from the fact that charging a bank of lithium batteries at the same time as they are discharging is a demonstrably poor idea there are also other
very important reasons not to engage in this practice.
For one thing, lithium-ion power banks were never designed to be permanently installed in an automotive environment where they are subject to constant vibration and shock that inherently causes stress to the cells in the unit just by itself without even
any charging or discharging going on.
Then factor in
@blink that you live in Singapore, a tropical climate that is always hot and humid. Heat is probably one of the most stressful things that can happen to lithium batteries, especially in the oven-like temperatures inside a car in a hot climate. So, if you permanently keep a powerbank connected to a charging circuit while it is at the same time discharging - which is something it was never ever designed to do and you do so inside the hot, vibrating, shock prone environment of an automobile, you are asking for trouble.
Fortunately, these types of fires and explosions are relatively rare but they do indeed happen on a regular enough basis to be of concern and this is the risk one is taking. Exploding lithium-ion battery cells have caused severe burns, loss of limbs, eyesight, and even deaths. Exploding or burning lithium-ion batteries have caused cars to burn up and houses to burn down.
So, I'll once again repeat your question along with my answer:
"
Can I use a normal powerbank for my purpose?"
"No".